|
9/14/2023 0 Comments Ns2 np3 valence shell group 5a
What is the most electronegative element in group 5A? The valence electron configuration of the group 5A elements is ns2np3. Five elements are less frequently so classified: carbon, aluminium, selenium, polonium, and astatine. How many of the elements of Group 5 are metalloids? The six commonly recognised metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium. As in group 14, the heavier group 15 elements form catenated compounds that contain only single bonds, whose stability decreases as we go down the group. 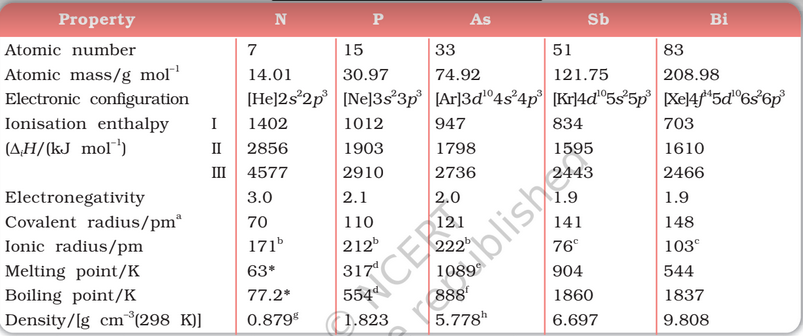
What is the heaviest metalloid in group 5A? The three heaviest pnicogens-arsenic, antimony, and bismuth-all have a metallic luster, but they are brittle (not ductile) and relatively poor electrical conductors. Is there a metalloid in Group 5a? Group 5A (or VA) of the periodic table are the pnictogens: the nonmetals nitrogen (N), and phosphorus (P), the metalloids arsenic (As) and antimony (Sb), and the metal bismuth (Bi).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
